A unique evolutionary cross-over has been discovered for the first time in parasites living in the marine herbivorous fish, the western buffalo bream, in a find which may reveal more about how different species of parasite were able to diverge and adapt to different digestive and immune systems over time.
The unique parasite was discovered in an expedition off the coast of Perth, Western Australia by a team of researchers led by Daniel Huston from The University of Queensland’s School of Biological Sciences and was identified as a brand new species of digenetic trematode – more commonly known as flukes – in the Gyliauchenidae family due to its unique living habitat.
While gyliauchenids are mostly found in the digestive tracts of tropical marine herbivorous fish from the Indo-Pacific such as rabbitfish, parrotfish and surgeonfish, they had never been found before this inside a fish like the western buffalo bream which comes from the kyphosid family, Huston told Lab Down Under.
Another family of trematodes, the Enenteridae, are more commonly found in fish like the western buffalo bream. Enenterids have both an oral and ventral sucker on their mouth and what would be their abdominal area while gyliauchenids have a sucker on their posterior and appear to have lost their oral sucker as they evolved over time.
Gyliauchenids effectively hang off their host’s intestinal wall and are thought to survive on a fluidic diet, either feeding off the mucosal liquid barrier of the intestines or off the host’s actual intestinal contents, called lumen. On the other hand, enenterids feed off blood and tissue and possibly the host’s lumen – although this has still yet to be confirmed by researchers – and are attached to the intestinal wall by their two suckers.
An unexplained divergence
Huston said that the divergence of these two families of flukes into different host fish was unusual, considering how closely related they were. Both come from a common ancestor – the parasite from which both evolved into over time.
“What we’re looking at is a situation where we have these different marine herbivorous fishes. This original group of parasites, the ancestor of the gyliauchenidae and the enenteridae, diverged and they diverged into different host groups,” he said.
There was no explanation for why the flukes established themselves in different families of host fish, Huston added.
“We don’t really understand why that would happen. Why would we see that specific divergence but there be no cross-over? You would just expect there to be a cross-over between those two groups but we don’t see it.”
This changed however in November 2017 when Huston and his team found the new species of gyliauchenid, now named Endochortophagus protoporus, in the western buffalo bream.
The conditions inside the intestines and immune system of these fishes may be a key to determining why there was such a clear cut division in hosts between the two trematode families, Huston said.
“There’s something unusual about that specific fish, something unusual about its internal physiology, something unusual about its ecology, or its immune system that has permitted the establishment of both gyliauchenids and enenterids in one host.
“A further study of that fish might reveal how that family of trematodes actually diverged which would be quite significant if we can figure that out. A lot of the time, we have no real understanding of what the drivers of speciation are.”
Speciation is the creation of new species over the course of evolution. It can be driven by a variety of factors ranging from specific physiological functions in the host to a simple repeated exposure to a parasite over time.
Another unanswered question is why a gyliauchenid had colonised western buffalo bream off of Western Australia but not from other locations where the same species of fish had been collected and examined.

“Every host is an island”
The trematodes themselves are incredibly diverse with 150 families and thousands of species. These species are mostly different because of the hosts they live in with the worms themselves being fairly similar physically.
“Every trematode has a very similar body plan because they don’t need to innovate much in terms of their morphology. Think about it like every host is an island. So you’ve got different species that have colonised every single island,” Huston said.
Some species of trematode, like human blood flukes or schistosomes, are harmful to the host while others such as the gyliauchenids may not have any negative effects at all.
Trematodes have a complex lifecycle with two or three different hosts. They start out hatching from eggs as larvae inside molluscs, taking control of their host’s reproductive system. After transferring the mollusc’s reproductive tissue into their own through clonal asexual reproduction, they produce swimming larvae called cercariae which look similar to tadpoles.
These cercaria escape from the mollusc and seek a second host which they infect in different ways and turn into waiting cysts called metacercariae. This intermediate host is then consumed by a third host such as a fish, into which the trematode hatches.
Maturing into adults, the flukes then sexually reproduce, producing eggs that pass out into the water through the fish’s faeces. These eggs hatch, turning into another form of the parasite called a miracidium which swims around the water using fine hairs, seeking a mollusc to take over. At this point, the cycle begins again.
“There are a huge variety of different lifecycle strategies that different lineages of trematodes use because they all have different definitive hosts, they all have different intermediate hosts, and so they have to use different strategies in order to reach the specific hosts that they need to complete their lifecycle,” Huston said.
An evolutionary arms race
Individual species of trematodes have adapted over millions of years to dodge the immune system of their specific host, investing a lot of energy to do so.
“It’s commonly referred to as an evolutionary arms race where the host’s immune system is trying to identify and kill the trematode and the trematode is coming up with various strategies to hide from the immune system or otherwise fool the immune system of the host,” Huston said.
Over many generations, evolution creates a parasite that is excellent at hiding from the immune system of one particular host but which would be quickly killed off by the immune system of another.
For this reason, the trematodes in these marine fishes would not survive in our own digestive tracts if anyone were to eat these fishes, Huston said.
“We’re so different, our immune systems are so different, our physiology is so different that they would have almost no chance of surviving within us for even a few hours.”
Probing the microbiome
With the discovery of this new species of trematode, Huston says the next step is to determine what makes the western buffalo bream so different from other kyphosids so that two families of fluke have been able to colonise it.
Researchers could examine the ecology of both gyliauchenids and enenterids within the host to find out exactly what they are doing in there. A possibility was that the flukes could have a unique relationship with the fish’s gut microbiome, Huston said.
“I think that it’s entirely possible that we’re looking at a situation where these supposed parasites – the enenterids and the gyliauchenids – actually have a completely different relationship with their host altogether. They might be consuming something else. They might be feeding on the host’s food or maybe they have some sort of association with the other endosymbionts within the host.”
Scientists currently do not know where these trematodes get their energy from. While certain substances have been identified within the digestive tracts of these parasites, it is difficult to tell whether they are only ingesting these compounds incidentally or actually absorbing them.
“I think the future is really revealing what the endosymbiotic community – all the different individual species that comprise the endosymbiotic community – and understanding from where these trematodes are actually deriving their nutrition,” Huston said.
Author’s note: If you enjoyed this article, you can follow Lab Down Under on Facebook or support me on Patreon. I also have my own personal Twitter account where I’ll be sharing my latest stories and any other items of interest. Finally, you can subscribe here to get my weekly blogs in your inbox.
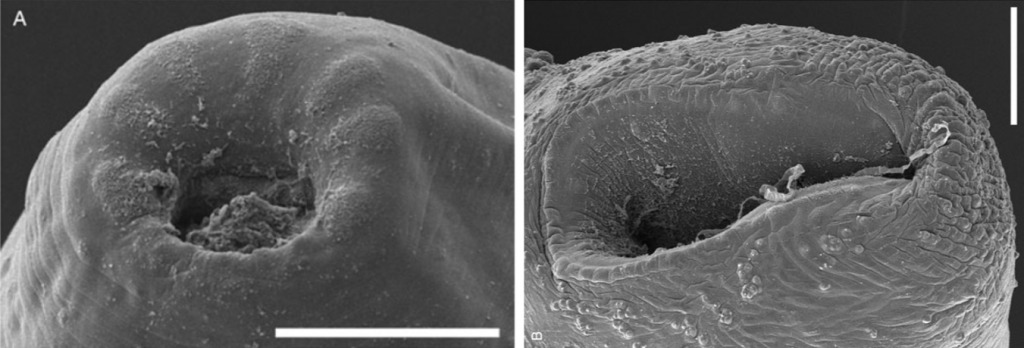
Featured image: Scanning electron micrographs of E. protoporus gen. nov., sp. nov. (A)
Anterior extremity, showing seven indistinct lobes with coarse tegument
surrounding mouth. (B) Posterior extremity and ventral sucker. Scale bars 100 μm, Daniel C. Huston, Cambridge University Press, 2019